The ultrafast light sources of Ultra at the Central Laser Facility have enabled a group of scientists led by Professor Peter Tonge from Stony Brook University, New York and Professor Steve Meech from the University of East Anglia to illustrate the interplay between chromophore dynamics and surrounding amino-acid residues - an interaction that underlies the process of fluorescent protein photochromism.
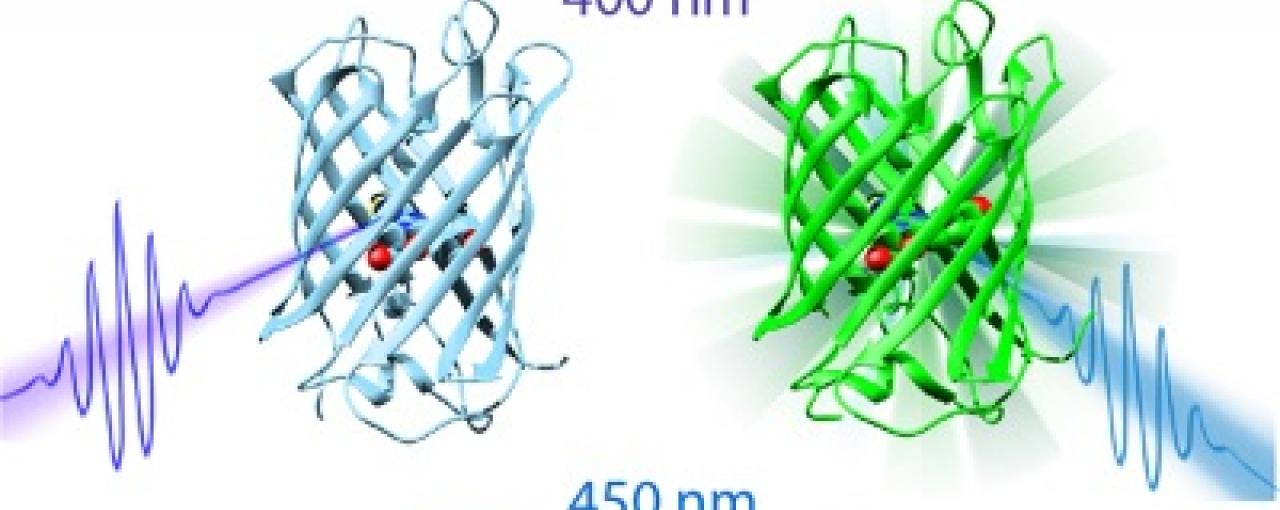
The team looked at a prototypical photochromic fluorescent protein called dronpa. Fluorescent proteins are special types of protein that possess a chemical compound called a fluorophore that can re-emit light after excitation. This is useful in nature because it allows for bioluminescence, which is used to attract mates or scare off predators. Photochromic fluorescence, however, is subtly different. It describes the process by which proteins undergo a reversible colour change when they are exposed to varying frequencies of light. Dronpa's light switch can be turned on or off; if blue light of 450 nm is absorbed by its fluorophore then it remains in an 'on' state. The protein's luminescence factory can then be turned off by stimulation with a UV wavelength of 400 nm.
Although scientists have previously highlighted the role of trans-to-cis isomerisation and proton transfer in dronpa photo activation, studies have usually been limited by factors such as spectral resolution or the accessible time range. Researchers have long struggled to unravel the actual mechanism by which dronpa is converted from the off to the on state.
However, using the ultrafast time-resolved infrared capabilities of the CLF's Ultra laser, the scientists were able not only to resolve the primary photochemical step in the on-off transition, but also to identify a series of ground-state structural transformations.
The diagram shows how the dronpa light switch can be switched on or off depending on the colour of light used to stimulate it (either 400nm or 450nm). Image: Stony Brook University.